Isingeniso
Ucwaningo lubonisa ukuthi ukwelashwa kwe-asidi yemfucumfucu yethusi ne-arsenic isebenzisa inqubo ehlanganisa imvula ye-sulfide, elandelwa ukungezwa kwe-gypsum kanye ne-electrochemical neutralization, ekugcineni okuholela ekukhishweni okuzinzile okuhlangabezana nezindinganiso zokulawula. Esinyathelweni semvula ye-sulfide, a i-sodium sulfide Isixazululo esine-mass concentration engu-26% sifakwa ngokuzenzakalelayo ku-asidi yemfucuza yethusi ne-arsenic ngaphansi kwezimo ezithile ze-asidi, okuholela ekwakhekeni kwe-CuS kanye ne-As2S3 precipitates ukuze kukhishwe ama-ion ethusi ne-arsenic esigabeni se-liquid. Ekukhiqizweni kwangempela, isisombululo se-sodium sulfide siqukethe amazinga aphezulu okungcola okungancibiliki, futhi lapho sichayeka emoyeni, kancane kancane siya oxidize senze i-thiosulfate. Ukwengeza, inezinga lokushisa eliphezulu le-crystallization, phakathi kwezinye izithiyo. Ngakho-ke, ukuthola i-ejenti entsha ye-sulfide esikhundleni se-sodium sulfide kungabaluleka kakhulu.

I-Sodium Hydrosulfide eqinile
Inqubo Isimiso
Ivinjelwa inqubo ekhona yokumuncwa ngokweqile kwe-H2S, ukwelashwa kwe-asidi yethusi ne-arsenic equkethe imfucuza kusetshenziswa. I-sodium hydrosulfide iqhutshwa ngokusekelwe kumathuluzi amanje kanye nokugeleza kwenqubo ye-sulfuric acid workshop's waste acid kanye namanzi angcolile. Le ndlela isebenzisa isisombululo esixubile se-sodium hydrosulfide ne-sodium sulfide ekwelapheni.

Ukugeleza kwenqubo
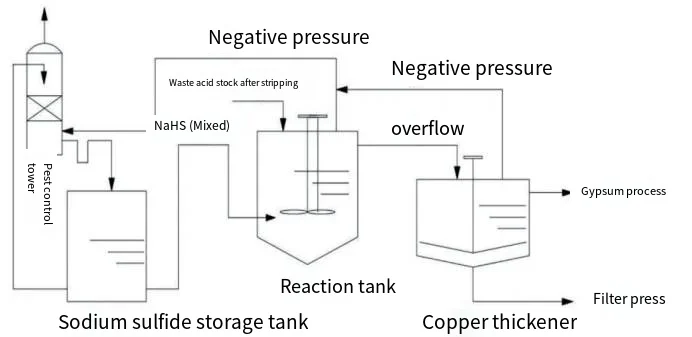
Umfanekiso we-1 ubonisa umdwebo wokugeleza kwenqubo yokwelashwa kwe-asidi engcolile yethusi ne-arsenic kusetshenziswa i-sodium hydrosulfide. Isixazululo se-asidi yemfucuza, ngemva kokususwa kwe-SO2 embhoshongweni wokuhlubula, singena ethangini lokusabela le-sodium hydrosulfide ukusuka phezulu. Isixazululo esixubile se-sodium hydrosulfide ne-sodium sulfide yengezwa kusukela phansi kwethangi lokusabela. Ngaphansi kwezimo ezine-acidic, igesi ye-H2S iyakhiqizwa futhi iyakhuphuka kancane kancane, isabela ngethusi ne-arsenic kusukela phezulu kuya phansi ukuze yakhe i-CuS ne-As2S3 precipitates. Lezi zimvula zibe sezichichima ku-thickener yethusi ukuze kunciphe. I-supernatant ithunyelwa kunqubo yokwelashwa kwe-gypsum, kuyilapho udaka olungaphansi luchithwa emanzini okuhlunga ngaphambi kokuthunyelwa ukuze kuqhutshekwe nokucubungula. Inani elincane le-H2S elingaphenduliwe lidonswa ipayipi elicindezelayo elingalungile embhoshongweni wokuqeda ubuthi, lapho limuncwa isixazululo esixubile esijikelezayo se-sodium hydrosulfide ne-sodium sulfide, iqinisekisa ukuthi ihlangabezana nezindinganiso zokukhipha.
Indlela Yokuhlola
Imodi yokukhiqiza eqhubekayo iyamukelwa, nezinye izinqubo nemingcele yokulawula ehlala ingashintshiwe. I-sodium hydrosulfide isabela ngokushesha kune-sodium sulfide; uma ukuhlushwa kuphakeme kakhulu, inani elikhulu le-hydrogen sulfide (H2S) likhiqizwa ngokushesha ngaphansi kwezimo ezine-asidi. Ngaphandle kokusabela ngenye yethusi ne-arsenic ku-asidi yemfucuza, i-hydrogen sulfide eningi ngokweqile izophunyuka endaweni ewuketshezi ngenxa yokuyaluza kwe-paddle enyakazayo futhi idonswe ukucindezela okungalungile kumbhoshongo wokuqeda ubuthi. Inqubo ekhona isebenzisa i-sodium sulfide ukuze imunce i-hydrogen sulfide eyeqile ekhiqizwa ngesikhathi sokusabela kwe-sulfide (Na2S + H2S = 2NaHS). Ngemuva kokusetshenziswa kwesivivinyo se-sodium hydrosulfide, ithangi lokugcina i-sodium sulfide liqukethe i-sodium hydrosulfide, enciphisa amandla ayo okumunca i-hydrogen sulfide eyeqile. Ukuze uvimbele ukumuncwa okungaphelele kwe-hydrogen sulfide, okungaholela ekukhishweni okungahambisani nemithetho evela embhoshongweni wokuqeda ubuthi, amanzi (acondensate asuka kumusi wokushisa wethangi le-sodium sulfide yokugcina) engezwa ethangini lokugcina i-sodium sulfide ngesikhathi sokuhlolwa ukuze kugcinwe ukugcwala kwesisombululo esixubile se-sodium sulfide ne-sodium hydrosulfide cishe ku-15%.
Isiphetho
Ngemva kokusebenzisa isixazululo esixubile se-sodium hydrosulfide kanye ne-sodium sulfide, izindleko ze-reagent yokwelapha i-cubic meter ye-waste acid ziyancipha zibe ngu-52.5% waleyo ye-sodium sulfide ehlanzekile, okuholela ezinzuzweni ezinkulu zezomnotho.
Ukusetshenziswa kwesisombululo esixubile se-sodium hydrosulfide ne-sodium sulfide akuzange kuthinte izinkomba zokukhishwa kwe-workshop; izindinganiso zokuchithwa kwamanzi angcolile zihlala zizinzile futhi zihambisana.
Ngenxa yokusetshenziswa okuxubile kwe-sodium sulfide ne-sodium hydrosulfide, kanye nenqubo ekhona yombhoshongo wokukhipha ubuthi engashintshiwe, azikho izimo zokungathobeli imithetho ekhishwayo evela embhoshongweni wokuqeda ubuthi.
- Okuqukethwe Okungahleliwe
- Okuqukethwe okushisayo
- Okuqukethwe kokubuyekeza okushisayo
- Isingeniso somkhiqizo wenkampani
- T-610 umqoqi we-Salicyl oxime acid Okuqukethwe okungu-3.5%
- I-sodium peroxide
- I-Citric acid-Ibanga Lokudla
- i-lithium Carbonates 99.5% Izinga Lebhethri noma 99.2% Ibanga lemboni 99%
- IGlycine Yezemithi Ephakathi Nekhwalithi Ephezulu 99%
- I-adipic acid 99% esetshenziswa njengempahla yenayiloni 66
- 1I-Sodium Cyanide Eyehlisiwe (CAS: 143-33-9) Yezimayini - Ikhwalithi Ephezulu Namanani Ancintisanayo
- 2I-Sodium Cyanide 98.3% CAS 143-33-9 I-NaCN i-ejenti yokugqoka igolide Ebalulekile Ezimbonini Zamakhemikhali Ezimayini
- 3Imithetho Emisha Yase-China Yokuthekelisa I-Sodium Cyanide Nesiqondiso Sabathengi Bamazwe Ngamazwe
- 4I-Sodium Cyanide (CAS: 143-33-9) Isitifiketi somsebenzisi wokugcina (inguqulo yesiShayina nesiNgisi)
- 5I-Cyanide Yamazwe Ngamazwe (i-Sodium cyanide) Ikhodi Yokuphatha - Amazinga Okwamukela Imayini Yegolide
- 6Imboni yaseShayina iSulfuric Acid 98%
- 7I-Anhydrous Oxalic acid 99.6% Ibanga Lemboni
- 1I-Sodium Cyanide 98.3% CAS 143-33-9 I-NaCN i-ejenti yokugqoka igolide Ebalulekile Ezimbonini Zamakhemikhali Ezimayini
- 2Ukuhlanzeka Okuphezulu · Ukusebenza Okuzinzile · Ukubuyisela Okuphakeme — i-sodium cyanide yokucwiliswa kwegolide yesimanje
- 3Izithasiselo Zokudla Okunomsoco I-Sarcosine Emluthayo 99% min
- 4Imithetho Yokungeniswa Kwe-Sodium Cyanide Nokuhambisana - Ukuqinisekisa Ukungeniswa Okuphephile Nokuthobelayo ePeru
- 5United ChemicalIthimba Labacwaningi Libonisa Igunya Ngemininingwane Eqhutshwa Ngedatha
- 6I-AuCyan™ High-Performance Sodium Cyanide | Ukuhlanzeka okungu-98.3% kokumbiwa kwegolide emhlabeni jikelele
- 7I-Digital Electronic Detonator (Isikhathi sokubambezeleka 0~ 16000ms)













Ukubonisana ngemiyalezo eku-inthanethi
Engeza amazwana: